Medical Technology
Smarter content solutions
Content Rules: 30+ years in
medical technology content optimization
Medical technology content optimization is essential to your business. Medtech teams handle complex, regulated, multilingual content. We replace legacy document workflows with structured content processes that speed delivery while maintaining quality and compliance.
Trusted by





















Strategic resources for medical technology
If your team is struggling to keep up with product development and regulatory expectations, it is time to rethink your approach.
AI and structured content change the game –enabling you to personalize your content at scale.
Most teams have squeezed as much optimization as possible out of their current processes. It is time to try a new approach. Structured content authoring is good for AI and enables scale, precision, and speed.
Structured content changes the game.
50%
Reduce the time and cost to translate content
Enable AI tools to create personalized content tailored to regulators, consumers,
and HCP
Improved operational efficiency and scalability
MedTech providers need modernize their content operations if they want to keep up.
Challenges facing the industry:
Regulatory pressure
Stay ahead of stricter global regulations by delivering clear, traceable, and transparent documentation that meets compliance standards.
Customer expectations
Modern users expect minimal friction when accessing product information. They want content that’s easy to find, relevant to them, and available in their preferred language.
Financial impact
The inability to manage content efficiently and translate content updates quickly can delay product launches, disrupt sales, and result in loss of market share.
Let Content Rules be your guide
We have traveled this road with many customers. Our experience and assistance can save you time and money.
For over 30 years, Content Rules has partnered with Medtech companies to reimagine and optimize their content strategies. We provide expert guidance and prepare you to tackle complex regulations, streamline workflows, and improve your bottom line.
Our services
When it comes to content operations, experience matters. At Content Rules, we’ve refined our consulting approach over decades of working with organizations facing challenges just like yours.
Why our expertise stands out:
- Tried and tested solutions: We know what works (and what doesn’t) because we’ve tested it all.
- Proactive issue resolution: By anticipating problems before they occur, we help you avoid costly setbacks.
- Faster implementation: Skip the steep learning curve. With years of experience guiding our process, we’ll get you on the path to success faster than starting from scratch.

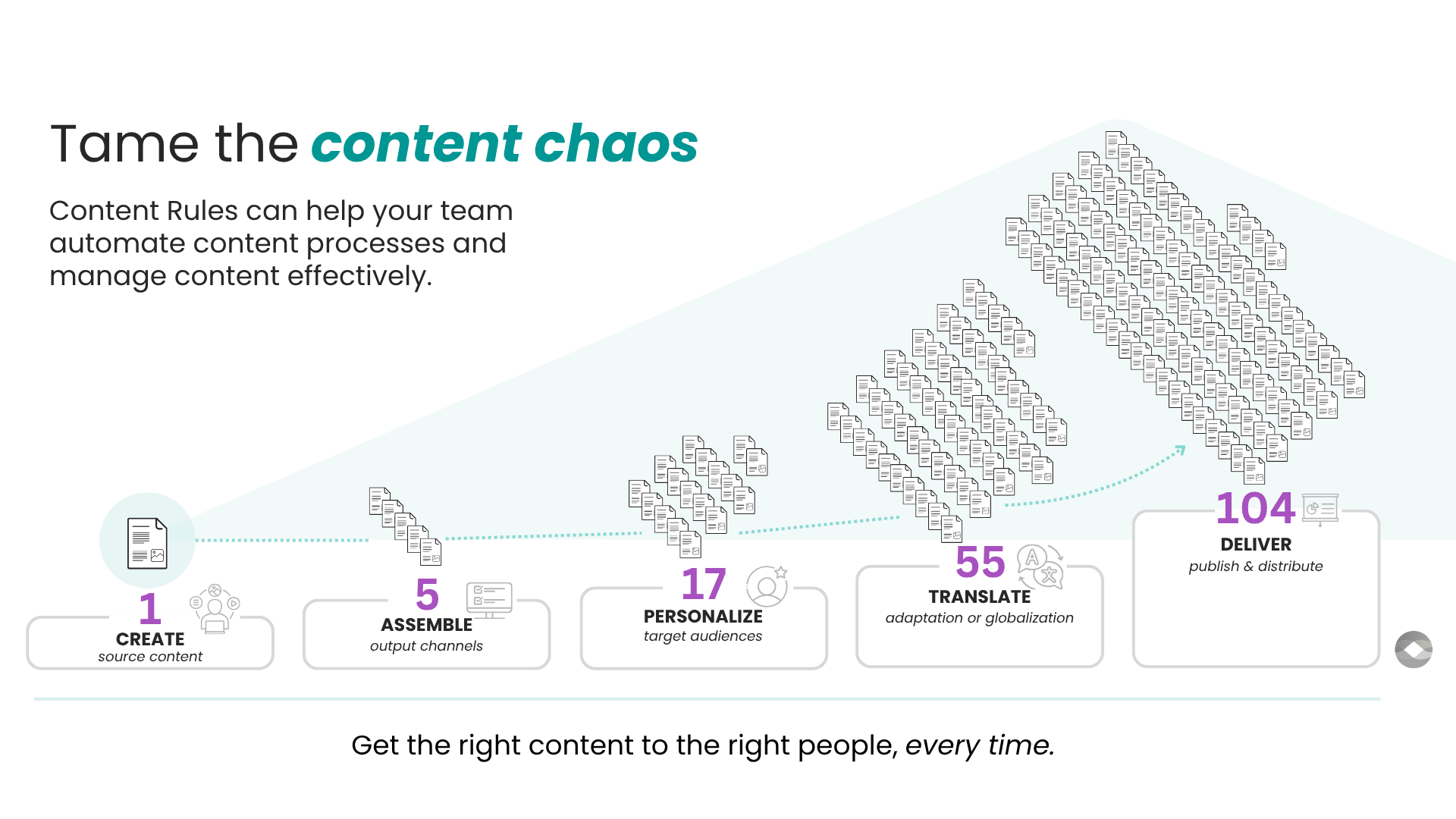
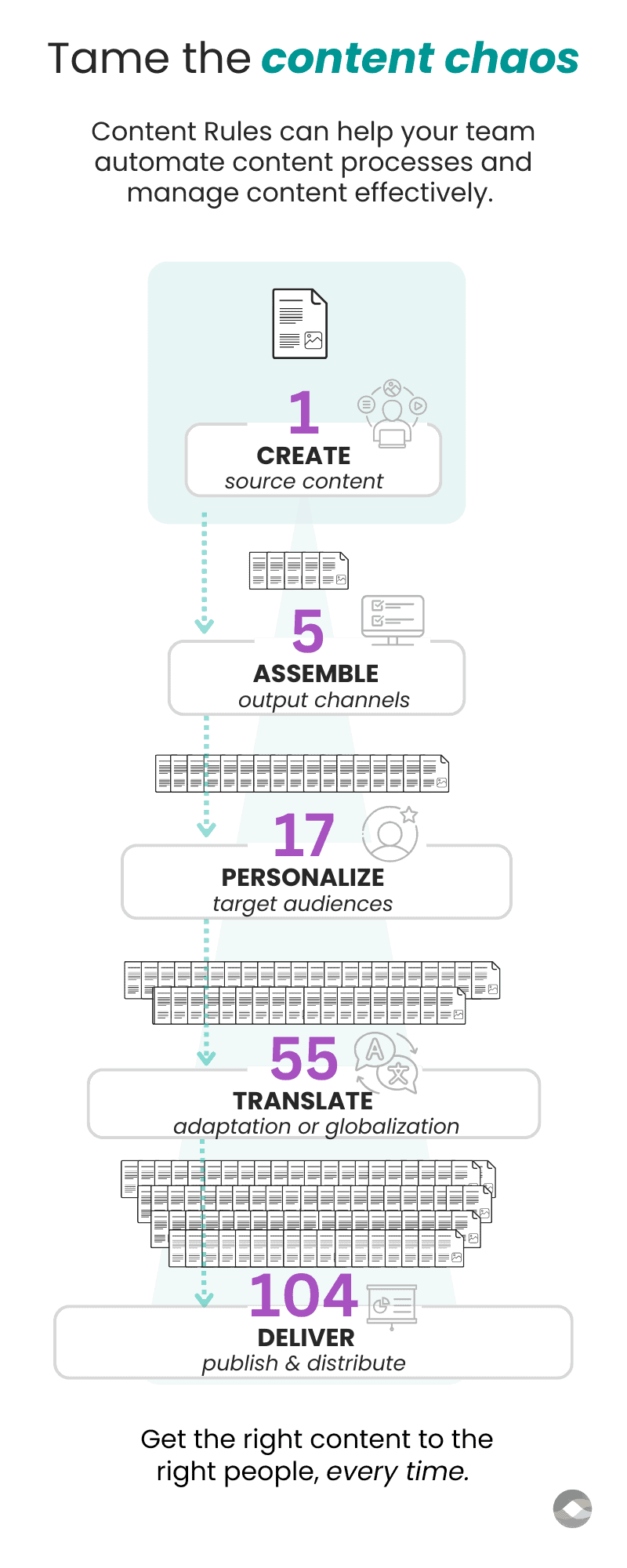
Medical technology content optimization in 3 simple steps
Content Rules is a content operations and transformation consultancy.
We help medical technology manufacturers and innovators to replace legacy document workflows with streamlined structured content solutions.
Medical technology companies produce large volumes of content throughout the device lifecycle. This content must meet regulatory requirements that vary by region. Much of it is translated into multiple languages. These factors pose unique challenges to speed, quality, and compliance.
1
Discover your needs
We assess your content systems, workflows, and goals to uncover areas for improvement. From regulatory challenges to localization gaps, we identify what’s holding you back and recommend strategies to break down the barriers.
2
Design & implement solutions
Our content experts can guide you through every step of your content journey, from strategy to execution. Solutions only work if your team can execute them. We prioritize practical, scalable solutions your team can sustain over time.
3
Achieve results & scale
Don’t get stuck! Build on your solution and extend its benefits across organizational functions. Empower your teams with efficient processes that deliver compliant, reusable, and scalable content faster than ever before.
Frequently asked questions
Medical technology companies that adopt structured content can improve user experience, reduce content creation time, and reduce compliance risk.
Structured content uses metadata, tagging, and reusable components to streamline version tracking, translations, and audit trails. This solution streamlines submissions to meet regional requirements.
Absolutely! Our globalization experts will optimize your content for global audiences. Experience shows that adopting structured content authoring significantly reduces translation time and costs.
Our expert team will guide you every step of the way, from diagnosing problems to defining solutions, selecting and implementing technology, and providing ongoing support and continuous improvement. We collaborate with your team to create solutions that work for you!
Transform your content & unlock opportunities
Medical technology companies that adopt structured content authoring gain a competitive edge by reducing risks, costs, and inefficiency.
Content Rules is here to guide you every step of the way.








