“We cannot change the authoring experience.”
This, more than anything else, is the message we used to be told (and still hear often) when we suggest a component-based structured authoring environment for medical writers at pharmaceutical companies. The authoring experience using Microsoft Word reigns supreme. Any changes to this interface are unacceptable.
Given the limitations of Microsoft Word, this requirement has previously been a real conundrum. Medical writers have been forced to use a tool that is no longer appropriate for the job.
We understand. Until recently, the interface for most XML-based authoring tools has been complicated. In fact, many of the tools on the market today are still quite complex.
These tools were created to support XML developers, technical writers, and content engineers. Consequently, these tools include dozens of useful features for programmers. Features that no medical writer will ever need to use. Let’s look at an example.
XML Without a Modern Authoring Tool
The following is an example of an SmPC in pure XML without the help of a Word-like XML authoring tool.

Who wants to deal with all that XML code on the screen when Word and Google Docs offer so much simplicity?
Modern XML Authoring
It took years and a lot of work to bury the complexity of XML-based structured authoring. However, there are tools on the market now that do just that. Rather than seeing and dealing with the complications of XML, the author sees a clean interface that is simple to use. These interfaces are very similar to Word or Google Docs.
Here is the same SmPC example using a modern, streamlined, and easy to use XML-based authoring tool.
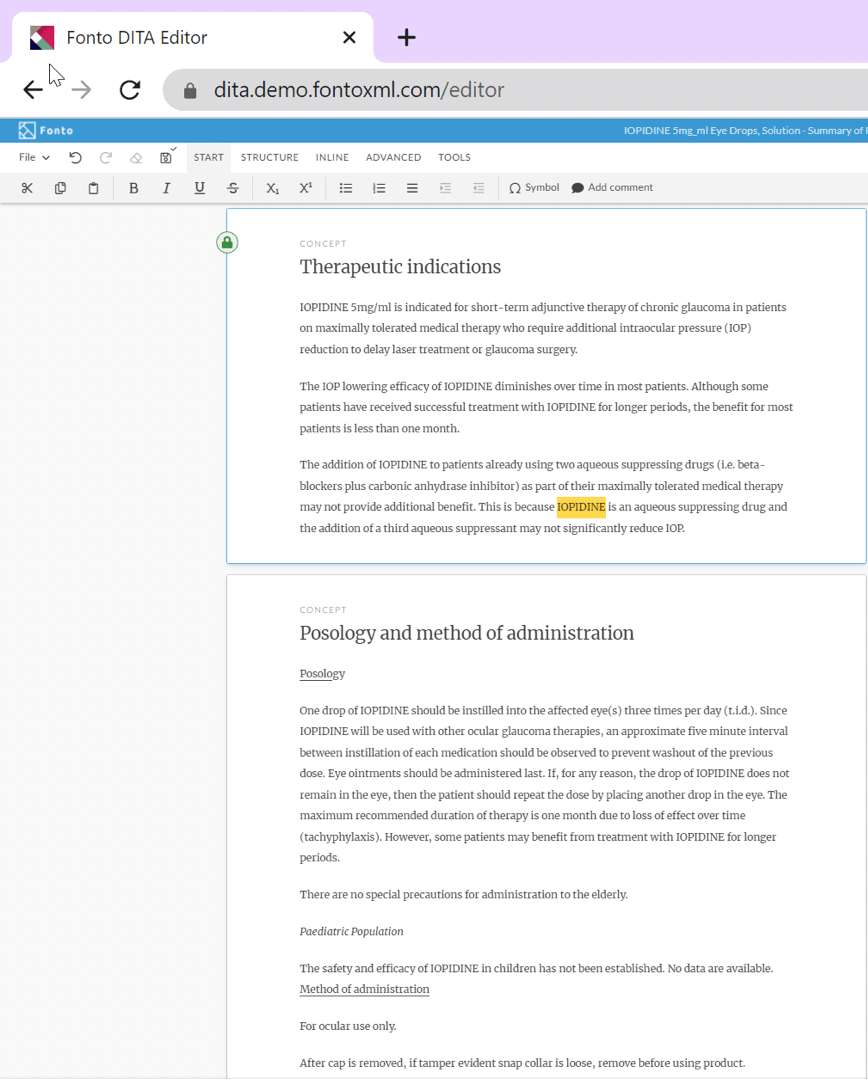
Under the technical covers, the authoring software performs the XML complexity, such as:
- Break content into components
- Tag the content appropriately
- Build the maps that define the order of the content in the output
- Filter content based on conditional logic
- Replace placeholders such as variables with data from a source of truth
All of this automation takes some initial configuration and is based on your content models, metadata design, and content reuse strategy. But the medical writers and authors and contributors never need encounter that level of complexity.
From Documents to Components
Another paradigm shift in XML-based authoring is the move from documents to components. In order to reuse small chunks of content, the content must be created and stored in those small chunks. We call the chunks components. XML-based authoring is often referred to as component-based authoring.
The shift from writing documents to writing content components can be difficult for some. In the past, many XML-based authoring tools displayed only a single component at a time. There was no way to see the entire “document” because there is no such thing as a document.
In component-based authoring, a document is a collection of components. A document is an output. It is assembled at the point of delivery, right before it is published.
Luckily, modern XML-based authoring tools such as Fonto XML show multiple components assembled in a document format. People accustomed to working in Microsoft Word or Google Docs see the entire document on the screen, and can easily scroll up or down as needed. Being able to use a simple authoring interface that retains the paradigm of document authoring for component-based XML content is a complete game changer. It is a major reason pharma companies are no longer locked into outmoded and insufficient tools for creating content.
Puzzle Solved
Imagine being able to create components that can be reused, mixed, and matched while not needing to learn a new tool to do so. This technological leap is the final jigsaw puzzle piece we have needed to revolutionize content creation in pharmaceutical companies.
Of course, there will be work to do in order to take full advantage of component-based structured authoring. There will be new systems to set up and configure, content that needs to be migrated, and training on how to create and use single-source content. One thing that will not require a lot of effort will be moving from Word to a new simple to use XML-based authoring interface.
At Content Rules, we have 27 years of experience helping customers design, build, and execute structured authoring solutions. We have worked with dozens of MedTech companies to help them implement the best solution for their content requirements. Drop us a line today to find out how we can help you solve the XML authoring conundrum, too.








